College of Liberal Arts & Sciences
5E40.03 - Batteries - Wet and Dry/Voltaic Pile
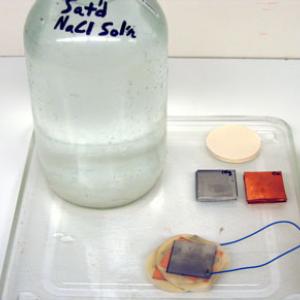
The batteries are displayed to show what they consist of and the different types that are available. No advanced preparation necessary other than routine cleaning.
A voltaic pile may be made using the copper and magnesium squares. Place a copper square on the bottom (this is one terminal of your voltaic pile), then a layer of filter paper, then a magnesium square, then a copper square directly on top of the magnesium square, and then another layer of filter paper. Continue this sequence until you have the desired number of layers in your pile, and end with a magnesium square on the top (this is the other terminal of your voltaic pile). Prep the filter paper with lemon juice or saturated salt water that has a small amount of vinegar added to it. Connect a Piezo buzzer or a voltmeter to the top magnesium plate and bottom copper plate of the pile for a measurement or audible indicator.
Take 5 of the pennies that have one face sanded off and place them all zinc side up. Soak 5 of the 1/2 inch square mat board pieces in saturated salt water that has a small amount of vinegar added to it. Place a soaked piece of mat board onto each of the zinc faces. Stack these penny-mat board pieces on top of each other, all with zinc sides facing up. Place an intact penny on top of the stack. Connect the red LED with the longer lead touching the intact penny and the other lead touching the bottom of the stack. Make sure the pennies don't touch each other or that the mat board doesn't drip as both of these events will short out the battery.
The large metal battery cell is made from an outer electrode of brass and an inner electrode of magnesium. You may use this as a show and tell demo or you can make this cell by putting 150 ml of any type of electrolyte in the cell and observe the voltage with a multimeter. Types of electrolytes that can be used include dilute acetic acid, any regular soda such as Coke or Mountain Dew, salt water, etc.
Several Weston standard cells are also available for demonstration.
- Wayne M. Saslow, "Voltaic Cells: The Good (Faraday), the Bad (Volta), and the Ugly (Galvani)", TPT, Vol. 59, #1, Jan. 2021, p. 22.
- John Lewis, "Simple Battery Experiment Reveals the Suprising Cost of Portable Electric Energy", TPT, Vol. 54, #4, Apr. 2016, p. 248.
- John Lewis, "Simple Take-Home Battery Experiment as Introduction to Electricity", TPT, Vol. 54, #3, Mar. 2016, p. 185.
- Mario Branca, Rossana G. Quidacciolu, and Isabella Soletta, "Big Pile or Small Pile?", TPT, Vol. 51, #7, Oct. 2013, p. 406.
- Stephanie V. Chasteen, N. Dennis Chasteen, and Paul Doherty, "The Salty Science of the Aluminum-Air Battery", TPT, Vol. 46, #9, Dec. 2008, p. 544.
- Adam Niculescu and Peter Martin, "Making and Evaluating an Electrical Battery", TPT, Vol. 44, #6, Sept. 2006, p. 343.
- Samaroo Deonarine, "Recharging the Battery Pack in a Camcorder", TPT, Vol. 41, #8, Nov. 2003, p. 497.
- Paul Changon, "What Do Battery Testers Test?", TPT, Vol. 34, #1, Jan. 1996, p. 16.
- Paul Hewitt, "Figuring Physics", TPT, Vol. 29, #3, Mar. 1991, p. 190.
- L. V. Hmurcik and J. P. Micinilio, "Contrasts Between Maximum Power Transfer and Maximum Efficiency", TPT, Vol. 24, #8, Nov. 1986, p. 492.
- H. Richard Crane, "What is the Secret of "Maintenance-Free" Car Batteries?", TPT, Vol. 23, #7, Oct. 1985, p. 432.
- Karl V. Kordesch and Klaus Tomantschger, "Primary Batteries", TPT, Vol. 19, #1, Jan. 1981, p. 12.
- R. D. Edge, "Current Electricity", TPT, Vol. 18, #6, Sept. 1980, p. 470.
- Fritz G. Will, "Sealed Batteries", TPT, Vol. 17, #8, Nov. 1979, p. 539.
- Thomas B. Greenslade Jr., "Voltaic Pile (Photo)", AJP, Vol. 77, #10, Oct. 2009, p. 889.
- W. M. Saslow, "How Batteries Discharge: A Simple Model", AJP, Vol. 76, #3, Mar. 2008, p. 218.
- Thomas B. Greenslade Jr., "Plunge Battery (Photo)", AJP, Vol. 73, #3, Mar. 2005, p. 233.
- Thomas B. Greenslade Jr., "Edison Cell (Photo)", AJP, Vol. 73, #2, Feb. 2005, p. 121.
- Wayne M. Saslow, "Voltaic Cells for Physicists: Two Surface Pumps and an Internal Resistance", AJP, Vol. 67, #7, July 1999, p. 574.
- Morris E. Wickliffe, "Electronic Precision Reference", AJP, Vol. 52, #1, Jan. 1984, p. 86.
- Dana Roberts, "How Batteries Work: A Gravitational Analog", AJP, Vol. 51, #9, Sep. 1983, p. 829.
- David H. Current, "Combinatorial Rules for Batteries", AJP, Vol. 47, #5, May 1979, p. 463.
- Karel Schell, "Optically Variable Ink", Physics Today, Vol. 78, #3, March 2025, p. 8.
- Jeffrey J. Richards, Julie B. Hipp, "The Black Powder Behind Battery Power", Physics Today, Vol. 77, #9, Sept. 2024, p. 26.
- David Kramer, "Fears of a Lithium Supply Crunch May Be Overblown", Physics Today, Vol. 74, #5, May 2021, p. 20.
- Matthew N. Eisler, "Cold War Computers, California Supercars, and the Pursuit of Lithium-Ion Power", Physics Today, Vol. 69, #9, Sept. 2016, p. 30.
- Sung Chang, "Better Batteries Through Architecture", Physics Today, Vol. 69, #9, Sept. 2016, p. 17.
- Richard J. Fitzgerald, "Surprising Stability for Sodium-Ion Batteries", Physics Today, Vol. 68, #8, Aug. 2015, p. 22.
- Johanna L. Miller, "A Battery Material Charges via an Unexpected Mechanism", Physics Today, Vol. #67, #9, Sept. 2014, p. 11.
- Johanna L. Miller, "Charged Polymers Form Unusual Nanostructures", Physics Today, Vol. 67, #8, Aug. 2014, p. 16.
- Larry C. Olsen, Peter Cabauy, and Bret J. Elkind, "Betavoltaic Power Sources", Physics Today, Vol. 65, #12, Dec. 2012, p. 35.
- Jacob Jorné, "Flow Batteries", American Scientist, Vol. 71, #5, Sept/Oct. 1983, p. 507.
- Ian Morse, "A Dead Battery Dilemma", Science, Vol. 372, # 6544, May 21, 2021, p. 780.
- Cyrus S. Rustomji, Yangyuchen Yang, Tae Kyountg Kim, Jimmy Mac, Young Jin Kim, Elizabeth Caldwell, Hywseung Chung, Y. Shirley Meng, "Liquified Gas Electrolytes for Electrochemical Energy Storage Devices", Science, Vol. 356, # 6345, June 30, 2017, p. 1351.
- C. Xia, C. Y. Kwok, and L. F. Nazar, "A High-Energy-Density Lithium-Oxygen Battery Based on a Reversible Four-Electron Conversion to Lithium Oxide", Science, Vol. 361, #6404, Aug. 2018, p. 777.
- Victoria K. Davis, Christopher M. Bates, Kaoru Omichi, Brett M. Savoie, Nebojša Momčilović, Qingmin Xu, William J. Wolf, Michael A. Webb, Keith J. Billings, Nam Hawn Chou, Selim Alayoglu, Ryan K. McKenney, Isabelle M. Darolles, Nanditha G. Nair, Adrian Hightower, Daniel Rosenberg, Musahid Ahmed, Christopher J. Brooks, Thomas F. Miller III, Robert H. Grubbs, and Simon C. Jones, "Room-Temperature Cycling of Metal Fluoride Electrodes: Liquid Electrolytes for High-Energy Fluoride Ion Cells", Science, Vol. 362, #6419, Dec. 2018, p. 1144.
- Lu Li, Swastik Basu, Yiping Wang, Zhizhong Chen, Prateek Hundekar, Baiwei Wang, Jian Shi, Yunfeng Shi, Shankar Narayanan, and Nikhil Koratkar, "Self-Heating–Induced Healing of Lithium Dendrites", Science, Vol. 359, #6383, Mar. 2018, p. 1513.
- Amartya Mukhopadhyay and Manoj K. Jangid, "Li Metal Battery, Heal Thyself", Science, Vol. 359, #6383, Mar. 2018, p. 1463.
- Shuting Feng, Jaclyn R. Lunger, Jeremiah A. Johnson, and Yang Shao-Horn, "Hot Lithium-Oxygen Batteries Charge Ahead: Molten Salt Electrolytes and Nickel Oxide-Based Electrodes Enable Four-Electron Transfer", Science, Vol. 361, #6404, Aug. 2018, p. 758.
- Sunghun Choi, Tae-woo Kwon, Ali Coskun, Jang Wook Choi, "Highly Elastic Binders Integrating Polyrotaxanes for Silicon Microparticle Anodes in Lithium Ion Batteries", Science, Vol. 357, #6348, July 2017, p. 279.
- Joseph F. Parker, Christopher N. Chervin, Irina R. Pala, Meinrad Machler, Michael F. Burz, Jeffrey W. Long, and Debra R. Rolison, "Rechargeable Nickel–3D Zinc Batteries: An Energy-Dense, Safer Alternative to Lithium-Ion", Science, Vol. 356, #6336, Apr. 2017, p. 415.
- Caroline Delbert, "The Holy Grail of Batteries is Here - Solid State Lithium Batteries Can Last 25 Years and Charge an Electric Vehicle in Minutes", Popular Mechanics, Jan. / Feb. 2022, p. 40.
- Caroline Delbert, "The Radioactive Diamond Battery That Will Run for 28,000 Years", Popular Mechanics, Sept./Oct. 2021, p. 20.
- Ezera Dyer, "5 Things To Know About Making Electric Car Batteries Better", Popular Mechanics, Vol. 193, #6, June 2016, p. 54.
- Kate Baggaley, "Make An LED Holiday Light Out Of Pennies", Popular Science, Vol. 285, #12, Dec. 2013, p. 91.
- Mike Walker, "Gray Matter: Batteries Out of Thin Air", Popular Science, Vol. 282, #2, Feb. 2010, p. 76.
- G. D. Freier and F. J. Anderson, "Ee-4", A Demonstration Handbook for Physics.
- G. D. Freier and F. J. Anderson, "Ee-3", A Demonstration Handbook for Physics.
- G. D. Freier and F. J. Anderson, "Ee-2", A Demonstration Handbook for Physics.
- Tik Liem, "The Coin Battery", Investigation to Science Inquiry, p. 262.
- George M. Hopkins, "Frictional Electricity", Experimental Science, p. 398.
- John Henry Pepper, "Electricity", Cyclopadic Science Simplified, p. 302.
- Rev. J. Lukin, The Boy Engineers, 1901, p. 324.
- Joseph H. Adams, "Cells and Batteries", Harper's Electricity Book For Boys, p. 12.
- Don Rathjen and Paul Doherty, "Saltwater Pentacell", Square Wheels, 2002, p. 85.
- Borislaw Bilash II and David Maiullo, "Soda Bottle Potential", A Demo a Day: A Year of Physics Demonstrations, p. 271.
- Rick Beyer, "Persian Power", The Greatest Science Stories Never Told, p. 4.
- "93. Making a Simple Battery: The Gerber Cell", Chemical Demonstrations Vol. I.
- "56. Exciting Electricity", The Ben Franklin Book of Easy & Incredible Experiments.
- Cash, Parker, and Taylor, "Battery Power", 175 More Science Experiments.
- Shar Levine and Leslie Johnstone, "Chapter 20: Power Arrangers", Everyday Science, p. 72.
- Douglas C. Myers, "Zap New Life Into Dead Nicads", Popular Electronics Magazine, July 1977.
- "How Batteries Supply Portable Electricity", Reader's Digest's How in the World, p. 12.
- Janice VanCleave, "Battery", 201 Awesome, Magical, Bizarre, & Incredible Experiments, p. 110.
- Ron Hipschman, "Hand Battery", Exploratorium Cookbook II, p. 91.1 - 91.2.
- Frederick C. Russell "That Black Box", Mechanix Illustrated, Aug. 1957, p. 146.
- "Volta's Pile Battery", New Illustrated Science & Invention Encyclopedia.
- Neil A. Downie, "Red-Hot Batteries", Ink Sandwiches, Electric Worms and 37 Other Experiments for Saturday Science, p. 301.
- Cy Tymony, "The Coin Battery", Sneaky Uses for Everyday Things, p. 24.
- Joey Green, "Battery Madness", The Mad Scientist Handbook, Vol. 2, p. 5.
- Bobby Mercer, "Three-Penny Battery", Junk Drawer Chemistry, 2016, p. 145 - 152.
- Grant Mellor, "Current Mysteries", Flying Tinsel, 1993, p. 80 - 82.
- Grant Mellor, "Inside a Flashlight 'Battery'", Flying Tinsel, 1993, p. 70 - 72.
- Grant Mellor, "Big Cell, No Shock", Flying Tinsel, 1993, p. 67 - 69.
- Grant Mellor, "A New Shock", Flying Tinsel, 1993, p. 62 - 66.
- Gerard L'E Turner, Nineteenth-Century Scientific Instruments, p. 197.
- Eveready Battery Company, "Eveready Carbon Zinc (Zn/MnO2) Application Manual", Energizer Battery Application Manual.
- Rudolf F. Graf, "Make Your Own Voltaic Pile", Safe and Simple Electrical Experiments, p. 77.
- Rudolf F. Graf, "Potato Polarity Indicator", Safe and Simple Electrical Experiments, p. 83.
- Herb Strongin, “Finding Out Which Battery Lasts Longest“, Science on a Shoestring, p. 162.
- Julius Sumner Miller, Q216 & A216, Millergrams II – Some More Enchanting Questions for Enquiring Minds, p. 66 & 114.
- Prof. Robert Griffith, "Electric Batteries", Boys' Useful Pastimes, p. 290.
- Joseph Frick, "# 278 - The Galvanic Pile", Physical Technics: Or Practical Instructions for Making Experiments in Physics and the Construction of Physical Apparatus with the Most Limmited Means, p. 315.
- "Battery sustained by bacteria", Teachers Clearinghouse for Science and Society Education Newsletter, Vol. XLI, #3, Fall 2022, p. 13
Disclaimer: These demonstrations are provided only for illustrative use by persons affiliated with The University of Iowa and only under the direction of a trained instructor or physicist. The University of Iowa is not responsible for demonstrations performed by those using their own equipment or who choose to use this reference material for their own purpose. The demonstrations included here are within the public domain and can be found in materials contained in libraries, bookstores, and through electronic sources. Performing all or any portion of any of these demonstrations, with or without revisions not depicted here entails inherent risks. These risks include, without limitation, bodily injury (and possibly death), including risks to health that may be temporary or permanent and that may exacerbate a pre-existing medical condition; and property loss or damage. Anyone performing any part of these demonstrations, even with revisions, knowingly and voluntarily assumes all risks associated with them.