College of Liberal Arts & Sciences
4C20.54 - Latent Heat of Fusion
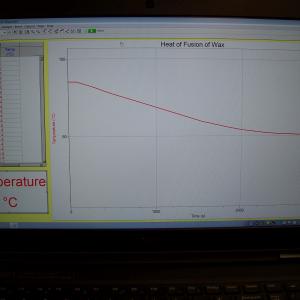
Place some wax into a beaker and melt it on a hot plate. Take the beaker off the hot plate and place a thermometer into the melted wax. Monitor the temperature as the wax cools and solidifies. A plateau in the cooling curve should be observed. It is good to melt the wax before class and let it cool to about 70 degrees C to start the class. The fusion plateau will be around 58 degrees C but the experiment will need to run at least 30 to 40 minutes to see good data.
The same experiment may be done using ice instead of wax. Make a small dry ice/alcohol bath in a Styrofoam cup. Put water into a test tube, insert the thermometer, and place into the dry ice/alcohol bath until all the water has frozen. In particular make sure that ice has developed around the thermometer. CAUTION: Do not leave the test tube in the dry ice/alcohol bath too long as a hard freeze will cause the test tube to break. The ice will be at a temperature below 0 degree C at this point. Wait till the temperature reads 0 degrees C before you proceed to the next step. Once the temperature is at 0 degrees C, place the test tube in a warm beaker of water and start the data collection. Note that the water level of the beaker of warm water should only come to a level that is below the tip of the thermometer in the test tube or convection from the sides of the test tube will give discrepant data results. Allow at least 20 minutes of class time for the data collection and results.
Other substances that would work are:
Coconut Oil, melts at 24 degrees C, (76 degrees F).
Hydrogenated Coconut Oil, melts at 36 to 40 degrees C, (97 to 104 degrees F).
Margarine or Crisco.
- James Lincoln, "Lab Activities on Temperature and Thermodynamics", TPT, Vol. 60, #1, Jan. 2022, p. 74.
- William A. (Toby) Dittrich, "Drop Tower Physics", TPT, Vol. 52, #7, Oct. 2014, p. 415.
- Trevor C. Lipscombre, Carl E. Mungan, "The Physics of Shot Towers", TPT, Vol. 50, April 2012, p. 218.
- Francis X. Hart, "A Microcomputer-Based Phase-Change Experiment", TPT, Vol. 36, # 2, Feb. 1998, p. 98.
- James L. Hunt, Tracy L. Tegart, "Measuring the Heats of Water", TPT, Vol. 32, Dec. 1994, p. 545.
- J. Harris, A. Ahlgren, "Apparatus-Lecture Demonstration and Laboratory. Classroom Techniques. Some Simple Experiments and Demonstrations", TPT, Vol. 4, #7, Oct. 1966, p. 314.
- Mahantappa S. Jogad, David Van Domelen, Gary Westfall, Walter Benenson, S. D. Mahanti, "Determination of Melting Temperature and Heat of Fusion of a Solid Using a Computer-interfaced Temperature Probe", AJP, Vol 70., #1, Jan 2002, p. 89
- Jack A. Soultes, "Improved Sophomore Experiment to Measure Latent Heat of Fusion", AJP, Vol. 35, #1, Oct. 1966, p. 23.
- Joseph Frick, "# 352 & 353 & 354 - Fusible Alloys & The Liberation of Caloric & Crystallization of Bismuth", Physical Technics: Or Practical Instructions for Making Experiments in Physics and the Construction of Physical Apparatus with the Most Limmited Means, p. 418 & 419.
Disclaimer: These demonstrations are provided only for illustrative use by persons affiliated with The University of Iowa and only under the direction of a trained instructor or physicist. The University of Iowa is not responsible for demonstrations performed by those using their own equipment or who choose to use this reference material for their own purpose. The demonstrations included here are within the public domain and can be found in materials contained in libraries, bookstores, and through electronic sources. Performing all or any portion of any of these demonstrations, with or without revisions not depicted here entails inherent risks. These risks include, without limitation, bodily injury (and possibly death), including risks to health that may be temporary or permanent and that may exacerbate a pre-existing medical condition; and property loss or damage. Anyone performing any part of these demonstrations, even with revisions, knowingly and voluntarily assumes all risks associated with them.